- The reaction of solid sodium hydrogencarbonate, NaHCO (s), with hydrochloric acid, HCl(aq), was investigated using the same mass of solid and a constant volume of different concentrations of acid. A data logger was used to measure the mass of the reaction mixture every five seconds.
The results were graphed.
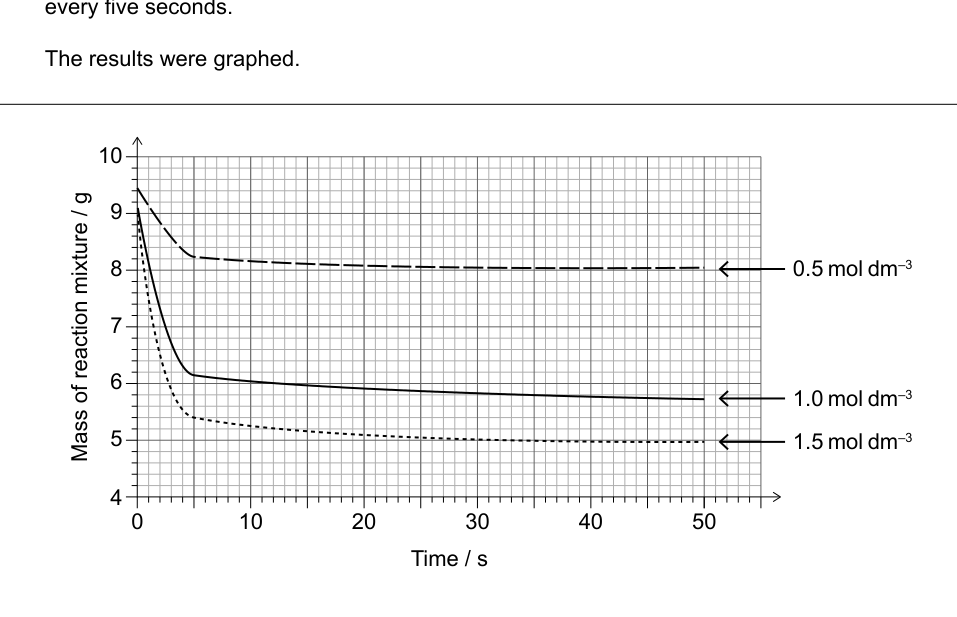
(a) Determine, by annotating the graph, the initial rate of release of CO (g), in g s, with the 1.5 mol dm HCl(aq). [2]
(b) Suggest, with a reason, whether NaHCO (s) or HCl(aq) was the limiting reagent in the reactions using 0.5 and 1.0 mol dm HCl(aq). [1]
(c) The expected (theoretical) mass of CO (g) produced in a complete reaction with 1.0 mol dm HCl(aq) is 4.40 g.
(i) Determine from the graph the actual mass, in g, of CO (g) produced. [1]
(ii) Calculate the percentage difference between the theoretical and actual mass of CO (g) produced. [1]
(iii) Although the same mass of NaHCO (s) and volume of HCl(aq) were used each time, the graphs begin from different points.
Suggest an error in the method that may have caused this. [1]