- An organic compound, A, has the following composition by mass when its only combustion products, carbon dioxide and water, are analysed.
| C / % | H / % |
|---|---|
| 71.93 | 12.10 |
(a) Outline why this compound is not a hydrocarbon. [1]
(b) Determine the empirical formula of A. [2]
(c) A sample of the vapour of A at 200.0°C, and 1.00 × 10⁵ Pa, has a density of 2.544 × 10³ g m⁻³.
Determine the molar mass and the molecular formula of A. [2]
(d) The infrared (IR) spectrum of A is shown below.
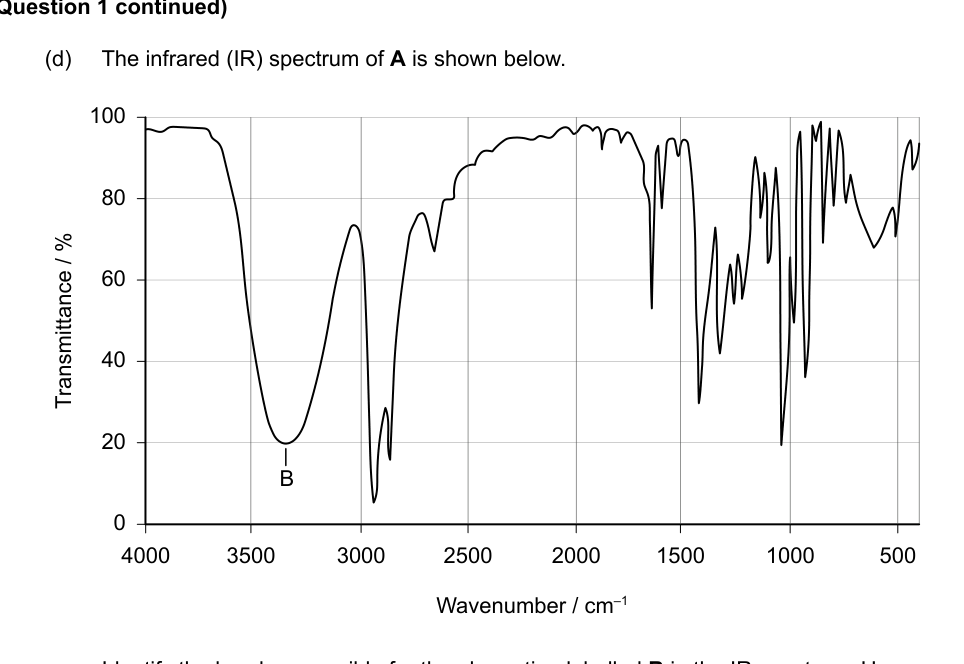
Identify the bond responsible for the absorption labelled B in the IR spectrum. Use section 26 of the data booklet. [1]
(e) A can be converted to compound E, which has a higher molecular mass, by heating it under reflux with acidified potassium dichromate(VI), K₂Cr₂O₇.
Identify one functional group present in E based on this information only. [1]
(f) Deduce a possible structural formula for A consistent with the evidence presented. [1]