- In the stratosphere, ozone is decomposed by ultraviolet radiation.
O₃ (g) → O₂ (g) + O(g)
(a) State the full electron configuration of an oxygen atom and the number of unpaired electrons in that atom. [2]
(b)
(i) Draw a Lewis (electron dot) structure for the ozone molecule. [1]
(ii) Deduce the formal charge on each of the three oxygen atoms by adding them to your Lewis (electron dot) structure in (b)(i). [2]
(iii) Predict the shape and bond angle of the ozone molecule. [2]
(iv) State the hybridization state of the central oxygen atom in ozone. [1]
(c) Suggest a value, in pm, for the bond lengths in the ozone molecule and explain your answer. Use section 10 of the data booklet. [2]
(d) Explain the dependence of the dissociation of diatomic oxygen, O₂, and ozone, O₃, on the wavelength of UV radiation. [2]
(e) The concentrations of ozone molecules and chlorine monoxide, ClO, free radicals were measured.
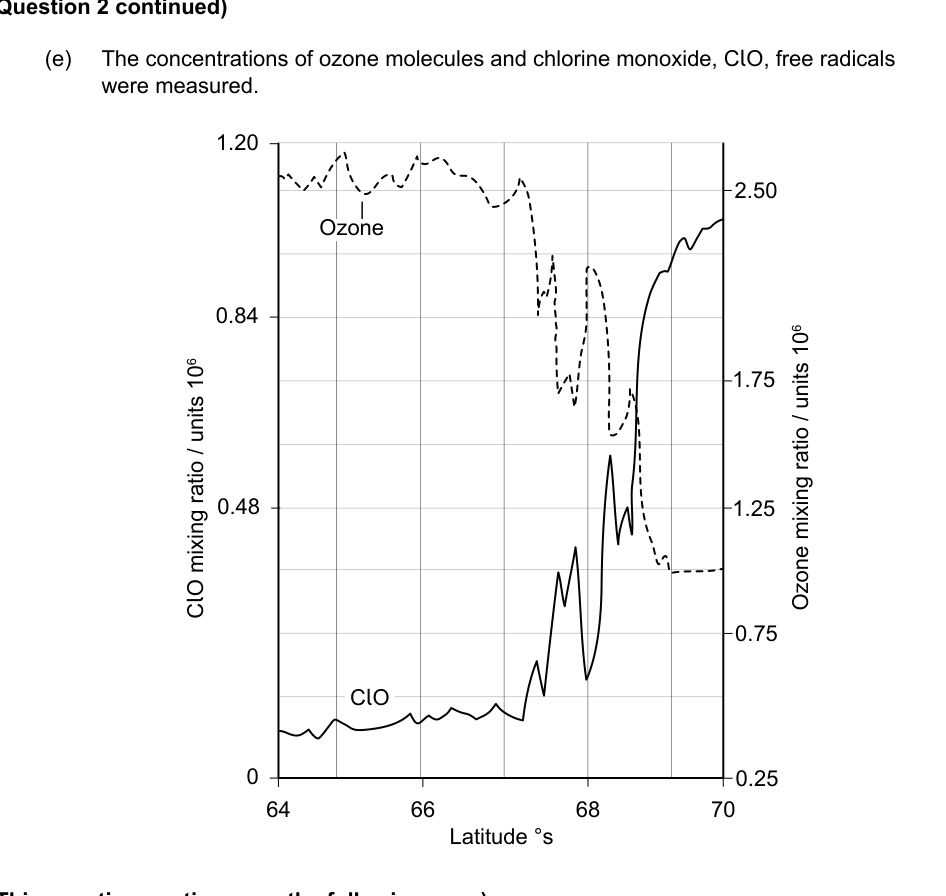
(i) Outline the relationship between the concentrations of ozone and ClO, free radicals. [1]
(ii) Comment, based on this graph, on the conclusion that the hole in the ozone layer is caused by ClO free radicals. [2]
(iii) When a chlorine emission spectrum is produced, there is a strong line at 453 nm. Determine the energy of the photon of light emitted in J, using sections 1 and 2 of the data booklet. [2]