- Ethenone, CH₂CO, is used in the synthesis of pharmaceutical compounds. structure of ethenone: H₂C=C=O
(a) Suggest why the compound is given this IUPAC name. [2]
(b) Compare and contrast the intermolecular forces that result in ethenone being less volatile than carbon dioxide. [2]
(c)
(i) Calculate the standard enthalpy change for this reaction. Use ΔH°f ethenone = –87.2 kJ mol⁻¹ and section 12 of the data booklet. [2]
(ii) Sketch the potential energy diagram for the thermal decomposition of propanone from (c)(i). Use the axis given and indicate both the enthalpy of reaction and the activation energy. [2]
(d) Ethenone can be converted to compound G, which reacts slowly with metal oxides when in aqueous solution. The mass spectrum of G is shown.
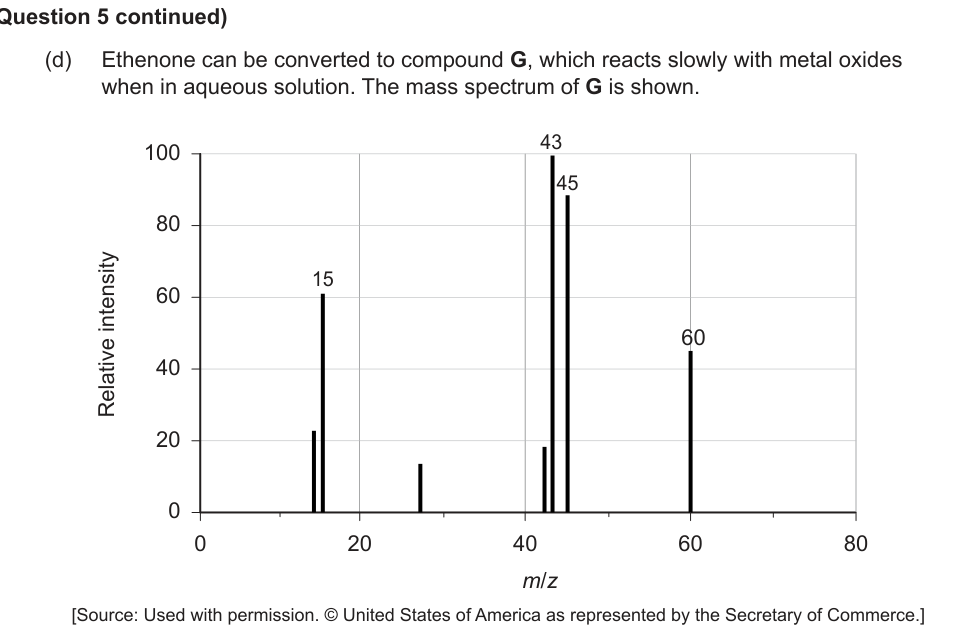
Deduce the identity of G giving two reasons based on the spectrum. [3]
(e) 10.0 cm³ of ethenone is mixed with 100 cm³ of oxygen and burnt completely.
CH₂CO (g) + 2O₂ (g) → 2CO₂ (g) + H₂O (l)
Determine the final volume of the gaseous mixture after the reaction mixture has returned to the original temperature and pressure. [2]
(f) Calculations often assume that real gases behave like ideal gases.
State one reason why gases such as carbon dioxide and ethenone become less ideal at higher pressures. [1]
(g) Propanone can be synthesised by the oxidation of propan‑2‑ol. Propan‑2‑ol can be synthesised from H by addition of water. reaction scheme showing H, water, propan‑2‑ol and propanone
Draw the structure of H. [1]
(h) Butan‑1‑ol can be produced from 1‑chlorobutane and sodium hydroxide.
(i) Identify the type of mechanism of this reaction. [1]
(ii) Explain the mechanism of the reaction using curly arrows to represent the movement of electron pairs. [3]
(i)
(i) Draw the stereoisomers of 2‑chlorobutane using wedge‑dash type representations. [1]
(ii) Outline how two enantiomers can be distinguished. [2]