- A student investigated the kinetics of the reaction between a dye, RCl, and aqueous sodium hydroxide. The dye has an intense blue colour that fades during the reaction.
RCl (aq) + OH⁻ (aq) → ROH (aq) + Cl⁻ (aq)
(Blue) (Colourless)
(a)
(i) Using the graph, determine the missing values from the table for experiment 3. Justify your answer. [3]
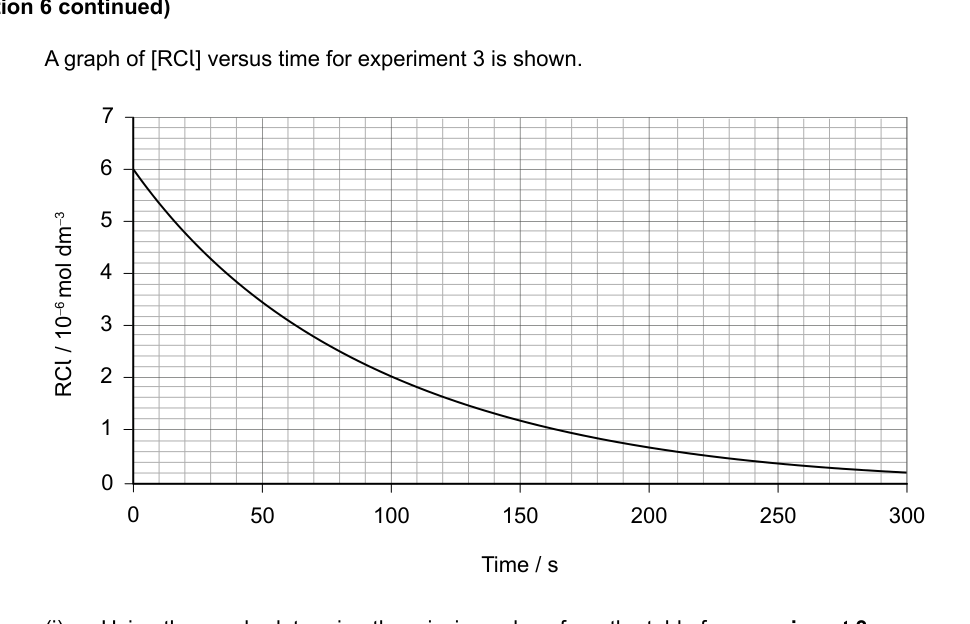
(ii) Deduce the order of the reaction with respect to each of the reactants. [2]
(iii) Calculate the rate constant with units at this temperature, using the data from experiment 1 in (a). [2]
(b) Explain why increasing the temperature increases the rate of a chemical reaction. [2]