- The structures of sodium bromide and sodium metal are shown below.
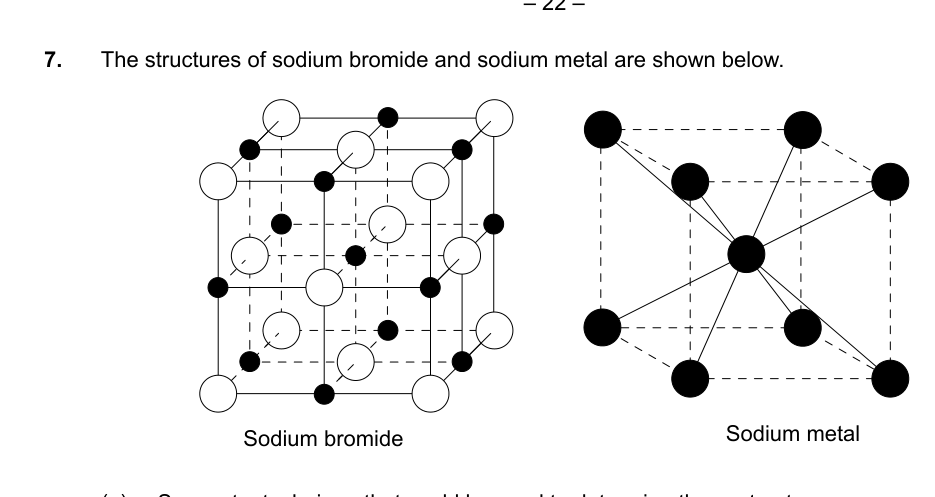
(a) Suggest a technique that could be used to determine these structures. [1]
(b) State and describe the bonding present in the two solid structures.
Na Br:
Na: [2]
(c) Write the half‑equations for the formation of the products at the positive electrode (anode) and negative electrode (cathode) when molten sodium bromide is electrolysed. [2]
(d) Determine the products formed at each electrode during the electrolysis of an aqueous solution of sodium bromide. Use section 24 in the data booklet. [2]
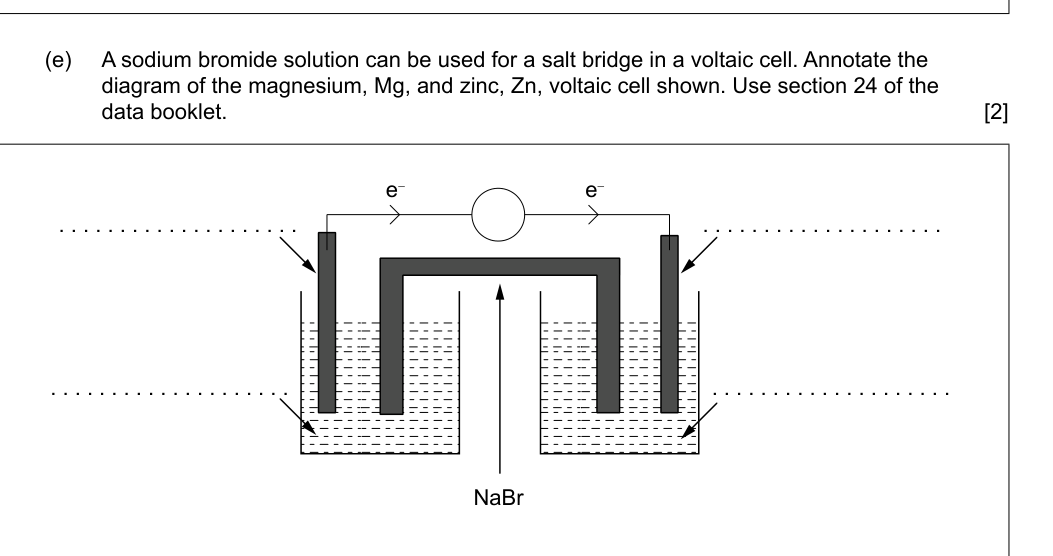
(e) A sodium bromide solution can be used for a salt bridge in a voltaic cell. Annotate the diagram of the magnesium, Mg, and zinc, Zn, voltaic cell shown. Use section 24 of the data booklet. [2]
(f) Determine the lattice enthalpy of sodium bromide using the data given below and sections 8 and 11 of the data booklet.
Na(s) → Na(g) ΔH = +107 kJ mol⁻¹
Br₂(l) → Br₂(g) ΔH = +30 kJ mol⁻¹
Na(s) + ½ Br₂(l) → NaBr(s) ΔH = –361.5 kJ mol⁻¹ [3]
(g) Calculate the enthalpy of solution of sodium bromide. Use your answer from (f) and section 20 of the data booklet. [2]
(h) Predict, giving a reason, the result of reacting aqueous sodium bromide separately with iodine and chlorine.
Iodine:
Chlorine: [2]