- Strychnine, C₂₁H₂₂N₂O₂ (Mr = 334.4), is a white crystalline solid obtained from plants.
(a) The formula of strychnine is:
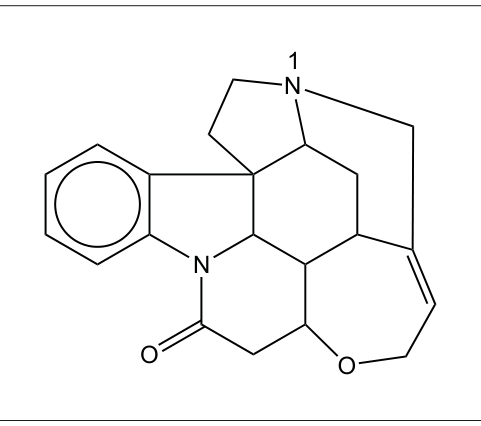
(i) State the name of the functional group containing the nitrogen atom labelled “1”. [1]
(ii) Outline how the functional group containing N₁ affects the pH when strychnine is dissolved in water. [1]
(iii) Circle a functional group that would react with bromine in the dark on the diagram in (a). [1]
(iv) State the number of rings in strychnine’s structure. [1]
(v) The strychnine structure contains chiral carbon atoms. Outline what is meant by this term. [1]
(b) 48.73 g of strychnine was converted into its sulfate by the reaction: 2 C₂₁H₂₂N₂O₂(aq) + H₂SO₄(aq) → (C₂₁H₂₃N₂O₂)₂SO₄(aq). Determine the percentage yield if 51.41 g of product was obtained. Use section 7 of the data booklet. [2]