- Crushed ice of mass 35 g at temperature –10 °C is placed in a warm room. The graph shows the variation of the temperature T of the ice with time t.
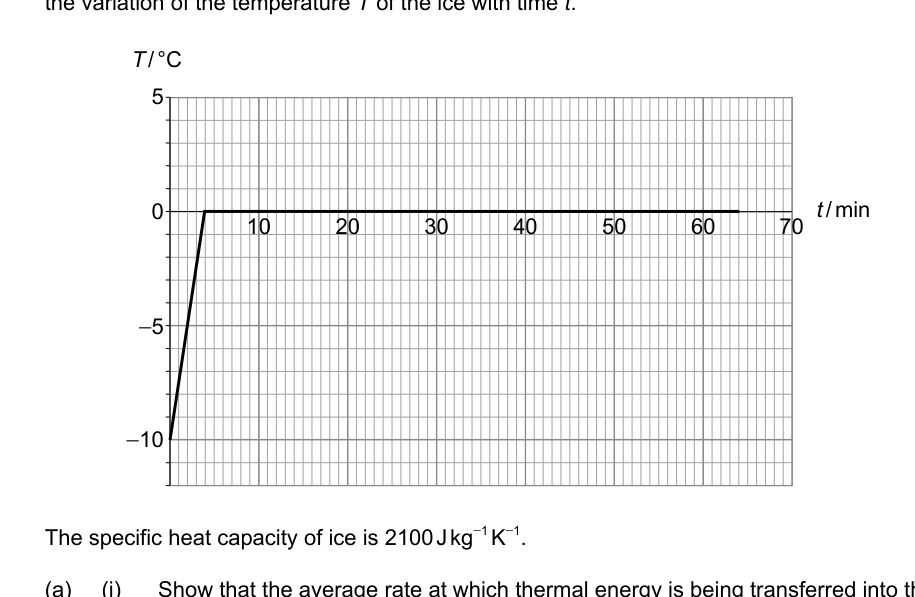
The specific heat capacity of ice is 2100 J kg⁻¹ K⁻¹.
(a) (i) Show that the average rate at which thermal energy is being transferred into the ice is about 3 W. [1]
(ii) Estimate the specific latent heat of fusion of ice. [2]
(b) Between 4 minutes and 64 minutes solid ice and liquid water coexist at 0 °C. Compare and contrast, during this time, the internal energy of solid ice to that of an equal mass of liquid water. [3]